At the beginning of 2020, the virus that causes COVID-19 was still known as “the novel coronavirus,” one that had never been seen before. Vaccines often take years to create and test, yet in a matter of months, scientists around the world had already developed and begun clinical trials on the first vaccines. How were they able to make such rapid progress?
That unprecedented speed was made possible by RNA vaccines, the same genetic technology that Deborah Fuller, PhD, a professor in the University of Washington Department of Microbiology, has devoted her career to. Now, she’s working on a next-generation vaccine that she thinks could be even better — and it could have a transformative impact far beyond this pandemic.
Creating a gene gun for vaccines
Fuller studied plant toxicology as an undergraduate, but as a graduate student, she realized it wasn’t where her heart was. While figuring out the next steps in her career, she started working at Agracetus, a biotech company.
Agracetus created transgenic crops using a handheld device called a gene gun, which shot DNA-coated gold particles into a plant’s cells. Fuller’s team was exploring whether the gene gun could also be used to inject DNA into the cells of live animals. The goal was to develop a new way to deliver gene therapy. The gene gun successfully micro-injected DNA into skin cells, but it also provoked a strong immune response. It seemed like a failure — until they were joined by a new investigator who had come from a vaccine laboratory.
“He took one look at the data and said, ‘Oh my gosh, it’s a vaccine,’” Fuller says.
But the scientific community was deeply skeptical. “In 1992, we presented our results at a conference,” says Fuller. “Everybody thought we were crazy.” A few of their peers saw promise in the new technology, and others were successfully delivering DNA in vivo with a needle and syringe, but most scientists didn’t think it could be scaled up for use on humans.
Initially, the naysayers were right. The first DNA and RNA vaccines, using a needle and syringe, were tested in the late 1990s and failed to induce good responses in human clinical trials. Although the gene gun was very effective in human clinical trials in 2000, interest in RNA and DNA vaccines had waned by then, and no one took much notice. But one company believed in the promise of DNA and RNA vaccines: Pfizer.
Pfizer acquired the gene gun technology and pursued DNA vaccines, but their efforts to make a disposable gun reduced its potency. Ultimately, they abandoned the technology in favor of mRNA vaccines formulated in lipid particles by a then little-known company called BioNTech. So, when Fuller asked to take the gene gun back for academic research in 2010, they agreed.
Now a professor of microbiology at UW Medicine, Fuller began developing a therapeutic HIV vaccine using the original gene gun, which showed very promising results in preclinical studies. She also had success delivering both seasonal vaccines and a “universal flu vaccine” that would protect against any influenza strain.
Then COVID-19 appeared — and Fuller and her team immediately saw the potential of DNA and RNA vaccines to battle a pandemic.
A third-generation vaccine
Pfizer went on to develop one of the world’s first COVID-19 mRNA (messenger RNA) vaccines. These vaccines work by teaching your cells how to build a harmless piece of “spike protein” that’s found on the surface of SARS-CoV-2, the virus that causes COVID-19. Your immune system recognizes that this foreign protein doesn’t belong and begins producing antibodies and activating immune cells. The mRNA doesn’t change or interact with your DNA in any way, and your cells quickly break down and dispose of the mRNA after they’ve built the spike protein. Now, if your immune system encounters SARS-CoV-2 in the future, it will “remember” it and be ready to fight off infection.
But SARS-CoV-2 is constantly mutating, creating new variants to outsmart our immune systems. Today, second-generation COVID-19 vaccines can be produced by tweaking the genetic formula to match emerging variants, but they’ll always be racing to catch up.
Instead, Fuller’s lab is working on an entirely different concept.
“We’re talking about a third-generation vaccine,” Fuller says. “Rather than chasing the variants, can we develop a universal vaccine where we can induce immunity against every variant out there and potential future variants?” In other words: a pan-coronavirus vaccine.
A pan-coronavirus vaccine would stimulate both antibodies and T-cells (a type of white blood cell that’s part of your immune system), because both are needed for the best protection from all variants. The initial shot would use DNA shared by all coronaviruses to give baseline immunity, and annual boosters would provide easily updated protection against specific variants, much like annual flu vaccines.
When scientists shared the genetic sequence of SARS-CoV-2 in early 2020, Jesse Erasmus, a Washington Research Foundation postdoctoral fellow in Fuller’s lab, quickly developed a replicon RNA vaccine, which makes copies of itself once inside the cell to stimulate strong immune responses using less RNA.
One major disadvantage of current COVID-19 vaccines is that they use a complex manufacturing process and special chemicals to coat the RNA in a lipid nanoparticle, which can lead to supply shortages and delays. Fuller and Erasmus’ vaccine uses an improved nanoparticle, called LION, that’s stable at room temperature, uses readily available materials and may require only one dose. Their LION vaccine is currently in Phase 1 clinical trials in India and is under consideration by the FDA to start a clinical trial in the U.S. in fall 2021.
“Vaccines are a public health endeavor. Everybody has to get together,” says Fuller. “And the only way that’s going to be accomplished is with a combination of different vaccines with different attributes to reach all corners of the world. It’s going to be a team of vaccines, and we think we have one part of that team.”
Beyond COVID-19
RNA vaccines are among our most powerful tools for ending the pandemic. But their uses extend far beyond COVID-19.
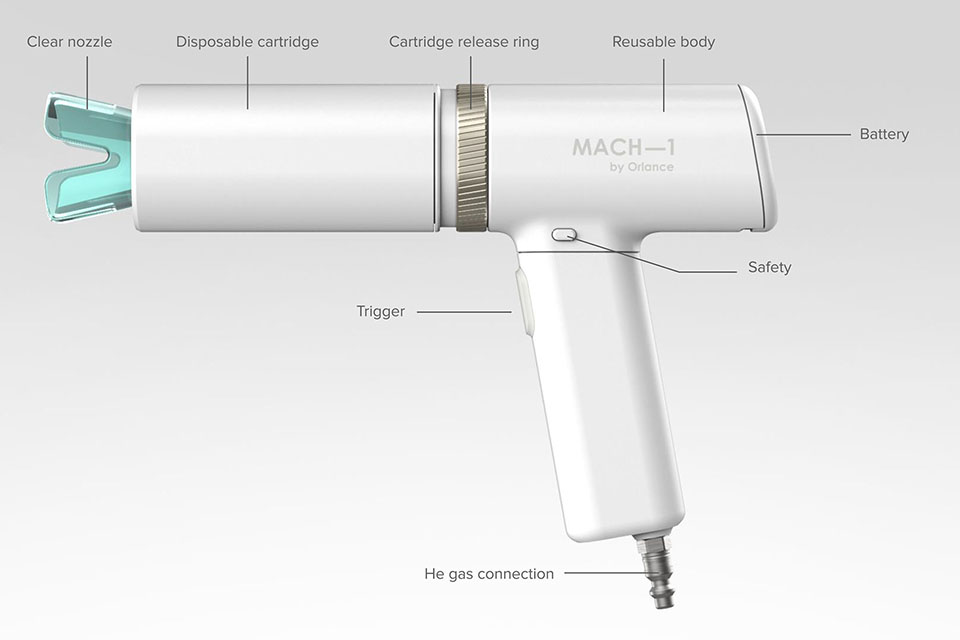 Since they’re highly customizable, RNA and DNA vaccines can be used for many immunologic diseases, including cancer, autoimmune diseases and infectious diseases. In addition, they could potentially replace vaccines currently in use that are less safe, cause adverse reactions, or are difficult to produce and distribute worldwide.
Since they’re highly customizable, RNA and DNA vaccines can be used for many immunologic diseases, including cancer, autoimmune diseases and infectious diseases. In addition, they could potentially replace vaccines currently in use that are less safe, cause adverse reactions, or are difficult to produce and distribute worldwide.
These vaccines could also have a global impact on improving healthcare equity. Because they are inexpensive to produce, stable at room temperature, storable for longer periods and may only require one dose, they can reach more people in more parts of the world.
And gene guns may even help protect our own communities from future outbreaks. Fuller’s lab is partnering with GE Global Health on a DARPA NOW U.S. government project that would stock nucleic vaccine kits in communities around the world. Each kit would have enough basic vaccine-making materials to quickly prepare RNA vaccines and treat the local community, stopping an outbreak before it can become a pandemic. The gene gun is being tested for inclusion in the kit as a way to easily administer vaccines.
When it comes to preventing the next pandemic, time is of the essence. But, Fuller says, the process of applying for grants is time-consuming, slowing down the research needed to accelerate our response to new COVID-19 variants and to combat the threat of future pandemics. That’s where philanthropy can make an important impact. Private funding would fast-track their work on universal vaccines for COVID-19, the flu and other diseases.
“Early in the COVID-19 pandemic, we were ready to respond, but there was no available funding to do the work,” says Fuller. “If not for a small grant from the Washington Research Foundation, we wouldn’t have been able to get our first COVID-19 vaccine off the ground and into clinical trials today.
“DNA and RNA vaccines are going to change the landscape, and donor support can get us there faster.”
With the potential to revolutionize the way we develop, produce and share vaccines, Fuller’s gene gun is right on target.
Written by Stephanie Perry
Top of page: Dr. Fuller in her lab with a prototype of the gene gun. Just above: Researchers are working on developing this gene gun to deliver micro-particles of a vaccine into the skin’s epidermis, which wouldn’t require a trained clinician and is stable at room temperature.
